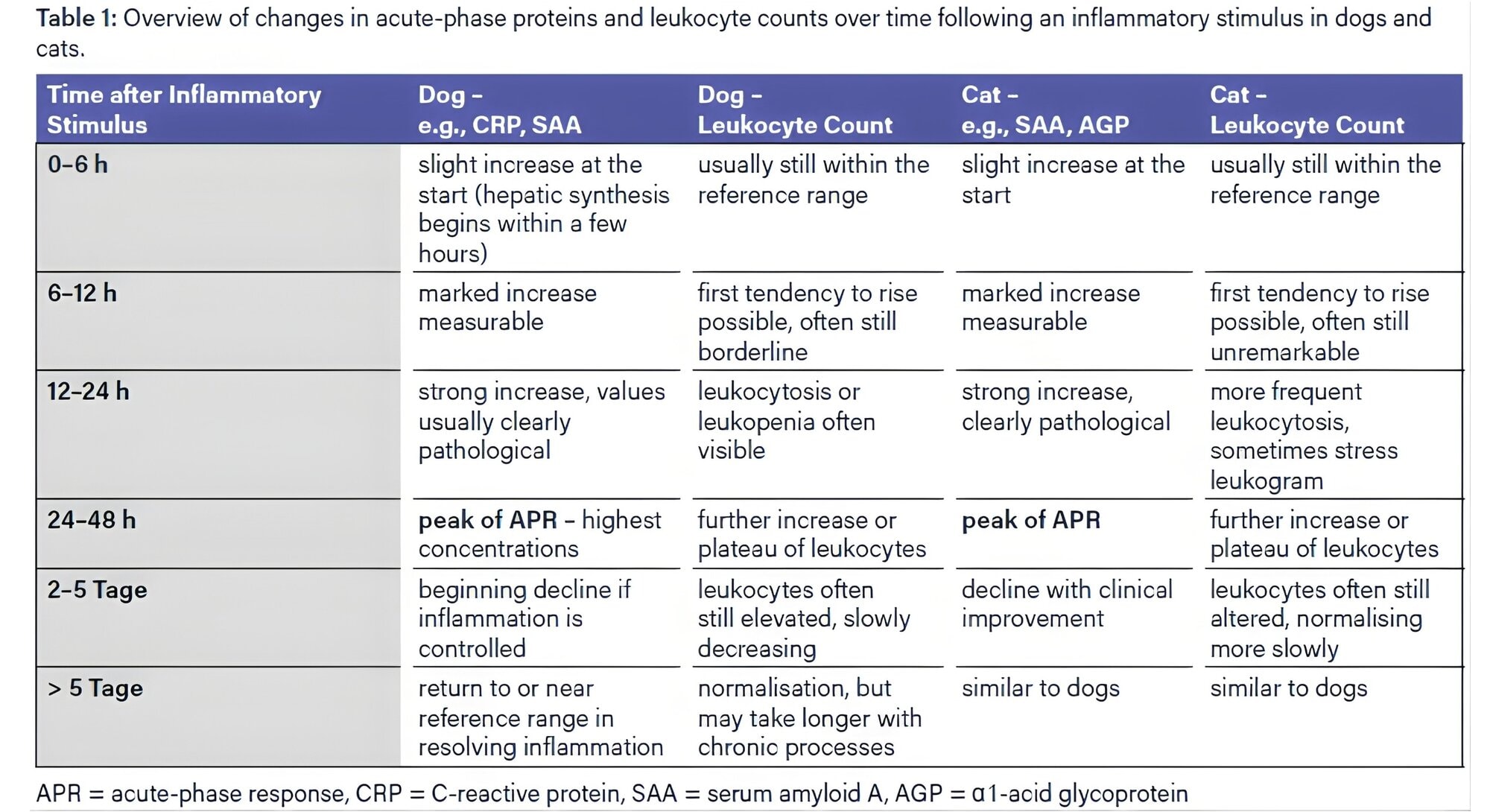
The acute-phase response (APR) is an early systemic reaction to inflammatory stimuli, infections, and tissue damage. The synthesis of specific acute-phase proteins (APPs) in the liver is regulated by proinflammatory cytokines (IL-6, IL-1β, TNF-α), with their concentrations in the blood rising or falling during an APR. APPs are valuable markers for detecting and monitoring the progression of inflammatory processes as well as neoplastic diseases. The APR occurs significantly earlier and more specifically than changes in leukocyte counts.
APPs are classified based on the magnitude of their response into major (10–100-fold increase), moderate (2–10-fold increase), and minor (<2-fold increase). Measurement of major APPs is particularly useful for early diagnosis and monitoring of diseases associated with an APR. In dogs, C-reactive protein (CRP) is the primary major APP, whereas in cats, serum amyloid A (SAA) predominates. Haptoglobin (Hp) and α1-acid glycoprotein (AGP) are considered moderate to minor responding APPs. Albumin, transferrin, and paraoxonase are negative APPs, whose concentrations typically decrease during an acute-phase response. The behaviour of the respective APPs during the APR is summarised in Table 1.
Dogs – CRP as the Gold Standard
As a major APP, C-reactive protein (CRP) increases within 4–24 hours following the triggering insult, rising up to 50–100-fold, reaches its peak after 1–2 days, and declines rapidly with effective therapy. Due to this dynamic behaviour, CRP is particularly well suited for early detection, monitoring disease progression, and assessing therapeutic success.
Elevated CRP levels are observed in a wide range of inflammatory and immune-mediated processes, including bacterial infections, parasitic diseases, autoimmune disorders, neoplasms, and post-traumatic or postoperative changes. In dogs with acute Babesia canis infection, CRP shows a strong correlation with clinical severity and haematological parameters.
Within the framework of antimicrobial stewardship, it has been shown that antibiotics can be discontinued once clinical improvement is observed and CRP concentrations have returned to normal, which significantly shortens the treatment duration for many diseases.
In systemic mycoses, such as pulmonary coccidioidomycosis, CRP in combination with haptoglobin (Hp) also demonstrated predictive value for remission. However, CRP can be elevated in the absence of inflammation, for example during extreme physical exertion or pregnancy, so its interpretation must always take the clinical context into account.
Acute-Phase Index (API) – a Combined Marker
Recent research has combined positive APPs (CRP, Hp) and negative APPs (albumin, optionally PON-1) into an acute-phase index (API). This index reflects the overall activity of inflammation. Dogs with
malignant tumours and a high API had a significantly poorer prognosis.
In chronic inflammatory diseases, such as canine leishmaniasis, CRP and Hp remain persistently elevated, while albumin and transferrin often decrease. Changes in the API correlate closely with treatment response and disease activity. Persistently high values indicate residual activity, co-infections, or treatment failure.
Cats – Focus on SAA and AGP
In cats, the dynamics and significance of APPs differ considerably from those in dogs. Serum amyloid A (SAA) is the most important major APP, while α1- acid glycoprotein (AGP) has particular diagnostic value in feline infectious peritonitis (FIP).
Serum Amyloid A (SAA)
SAA responds very early and sensitively, reaching high concentrations quickly, making it suitable for both early diagnosis and prognostic assessment. A rapid decline indicates a good response to therapy, while a stagnating value suggests persistent inflammation or secondary infection. Modern turbidimetric assays using monoclonal antibodies provide high diagnostic precision. Further studies demonstrate the usefulness of SAA, especially in bacterial infections such as pyelonephritis.
Alpha-1-acid Glycoprotein (AGP)
AGP is a moderately rising APP with high clinical relevance for FIP (Fig. 1). Elevated AGP serum levels support the presumptive diagnosis when interpreted alongside other findings. In particular, during antiviral therapy, AGP shows dynamic changes.
Haptoglobin (Hp)
In both dogs and cats, Hp is a moderate acute-phase protein synthesised in the liver. Its primary biological function is the high-affinity binding of free haemoglobin (Hb) from lysed erythrocytes, which reduces oxidative tissue damage and prevents the loss of Hb-bound iron. During acute inflammatory processes, both species exhibit a less pronounced and delayed increase in Hp concentration compared with major acute-phase proteins such as SAA or CRP. As in other mammals, intravascular haemolysis can lead to a decrease in haptoglobin concentration because the protein is rapidly consumed through binding large amounts of free haemoglobin.
Negative Acute-Phase Proteins
Albumin
Albumin decreases due to the redistribution of amino acids for the synthesis of positive APPs and increased capillary permeability. It is a valuable indicator of systemic inflammation but must be interpreted in the context of hydration status, protein loss, and liver function. In dogs, albumin is included in the calculation of the acute-phase index (API).
Transferrin
Transferrin, an iron-binding transport protein, decreases during the acute-phase response to reduce iron availability for microorganisms. In dogs, a marked decrease in transferrin has been observed during bacterial infections. In cats, a significant decline has also been documented in cases of chronic inflammation.
Acute-Phase Proteins in FIP
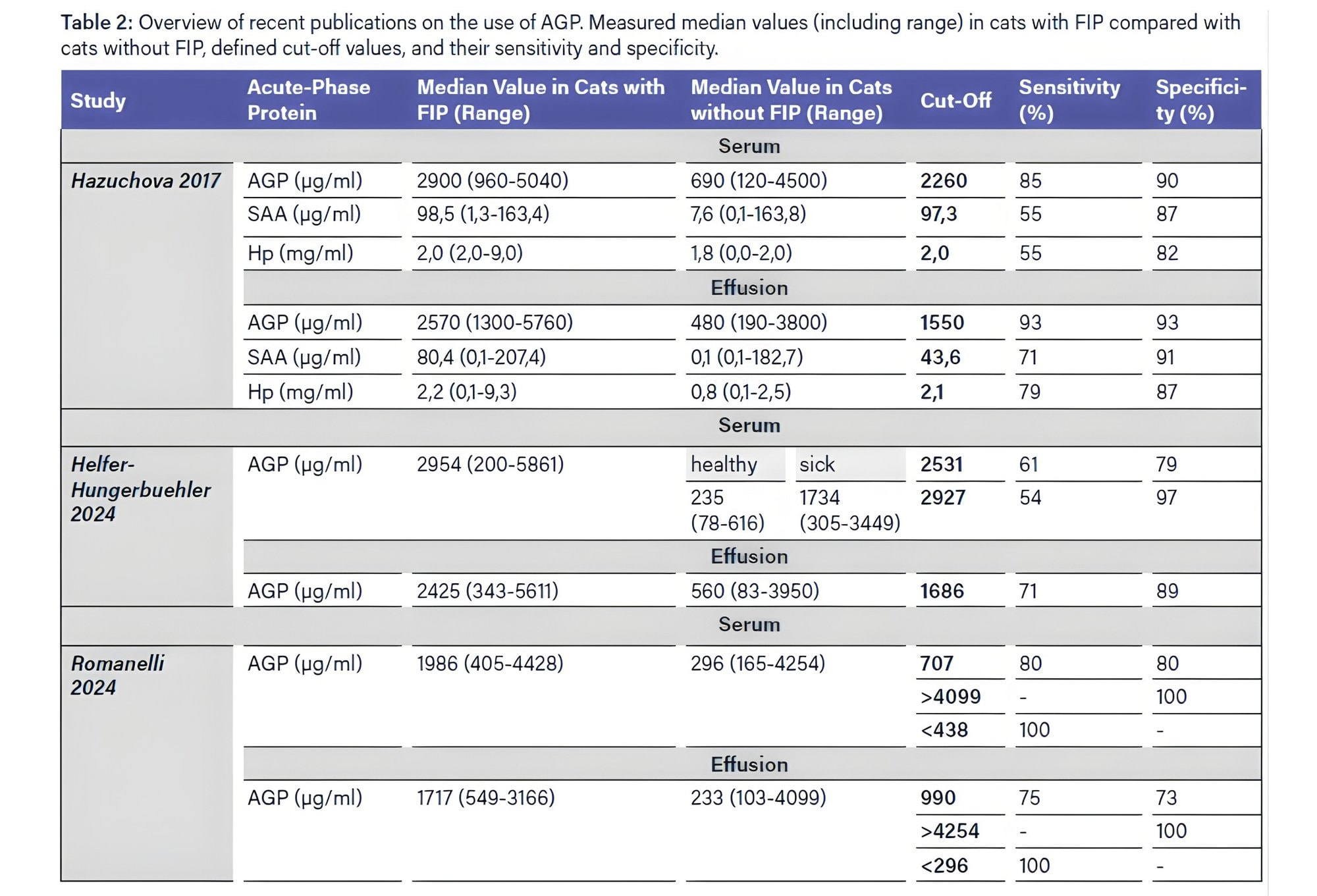
Feline infectious peritonitis (FIP) is an inflammatory disease that is generally associated with an increase in acute-phase proteins. Studies have shown that measuring AGP in effusions is the most informative method for distinguishing between cats with and without FIP. Different cut-off ranges with varying sensitivity and specificity have been defined (Table 2). Some of these cut-offs, for example in Helfer- Hungerbühler et al. (AGP > 2927), exhibit high specificity (97 %) and can therefore be strongly indicative of FIP. However, due to the relatively low sensitivity (54 %), almost half of the cats with FIP may not be detected. It should also be noted that APPs can increase in other diseases. Cats with a septic abdomen or disseminated neoplasms often show AGP concentrations similar to those observed in cats with FIP. Therefore, complementary cytological and bacteriological examinations are important to exclude differential diagnoses.
Measurement of AGP alone is insufficient for a definitive diagnosis and should be considered as one of many components in the diagnostic process. AGP may also play an important role in therapy monitoring for cats with FIP. During treatment, AGP gradually decreases, although more slowly than SAA. This is likely due to the longer half-life of AGP, which means that AGP concentrations on day 2 of FIP therapy can be higher than before treatment began. A significant decline in AGP was observed from day 7 after the start of therapy. By day 28, AGP levels were within the normal range in almost all cats (Helfer-Hungerbuehler 2024: 17 of 18 cats, Zuzzi- Krebitz 2024: 37 of 39 cats), making it a reliable parameter for monitoring treatment success. In contrast, SAA showed a significant decline already by day 2, and most cats reached (almost) normal SAA concentrations within 4–7 days. Addie and colleagues (2022) use AGP as a marker to differentiate between remission and recovery. Recovery refers to complete healing from FIP, while remission is defined as an intermediate stage between recovery and death, still carrying a risk of relapse. Cats that fully recovered showed AGP values within the normal range, whereas cats in
remission exhibited elevated AGP levels. Therefore, a rise in AGP could also indicate a potential FIP relapse.
Dr. Ruth Klein, Katharina Buchta
FURTHER LITERATURE Malin K, Witkowska-Piłaszewicz O. C-Reactive Protein as a Diagnostic Marker in Dogs: A Review. Animals (Basel). 2022 Oct 21;12(20):2888. doi: 10.3390/ani12202888. von Hohnhorst IM, Moritz A, Eisenecker CM, Strube C, Rodjana KE, Müller E, Schäfer I. Impact of levels of parasitemia and antibodies, acutephase proteins, as well as stays abroad on hematological and biochemical parameters in 342 dogs with acute Babesia canis infection. Parasit Vectors. 2025 Aug 15;18(1):347. doi: 10.1186/s13071-025-06997-4. Glick A, Jaffey JA, Kreisler R, Hanzlicek AS, Ringold R. Serum C-reactive protein and haptoglobin decrease in the first three months of treatment and relative change in haptoglobin predict remission in dogs with pulmonary coccidioidomycosis. J Am Vet Med Assoc. 2024 Jun 19;262(9):1222-1230. doi: 10.2460/javma.24.05.0296. Baldin M, Gelain ME, Marolato G, Bedin S, Berlanda M, Zanetti M, Torrigiani F, Giordano A, Moretti P, Scavone D, Bonsembiante F. Beyond Individual Acute Phase Protein Assessments: Introducing the Acute Phase Index (API) as a Prognostic Indicator in Dogs with Malignant Neoplasia. Vet Sci. 2025 Jun 1;12(6):533. doi: 10.3390/vetsci12060533. Fernandes Rodrigues N, Giraud L, Bolen G, Fastrès A, Clercx C, Gommeren K, Billen F. Antimicrobial discontinuation in dogs with acute aspiration pneumonia based on clinical improvement and normalization of C-reactive protein concentration. J Vet Intern Med. 2022 May;36(3):1082- 1088. doi: 10.1111/jvim.16405. Viitanen SJ, Lappalainen AK, Christensen MB, Sankari S, Rajamäki MM. The Utility of Acute-Phase Proteins in the Assessment of Treatment Response in Dogs With Bacterial Pneumonia. J Vet Intern Med. 2017 Jan;31(1):124-133. doi: 10.1111/jvim.14631. Yuki M, Inden T, Hirano T, Naito E, Taira H, Yokota S, Narita M. Comparison of polyclonal and monoclonal antibody assays for serum amyloid A in cats: a study based on an automated turbidimetric immunoassay in a primary care veterinary hospital. Am J Vet Res. 2024 Jul 12;85(9):ajvr.24.03.0067. doi: 10.2460/ajvr.24.03.0067. Waugh EM, Haining H, Harvie J, Ridyard AE, Eckersall PD. Validation of an automated immunoturbidimetric assay for feline serum amyloid A. BMC Vet Res. 2022 Sep 28;18(1):359. doi: 10.1186/s12917-022-03456-5. Kurtz M, Pey PBM, Mortier J, Manassero M, Da Riz F, Canonne-Guibert M, Maurey C, Benchekroun G. Usefulness of serum amyloid A for the diagnosis of pyelonephritis in cats: A prospective evaluation. J Vet Intern Med. 2024 May-Jun;38(3):1542-1552. doi: 10.1111/jvim.17082. Helfer-Hungerbuehler AK, Spiri AM, Meili T, Riond B, Krentz D, Zwicklbauer K, Buchta K, Zuzzi-Krebitz AM, Hartmann K, Hofmann-Lehmann R, Meli ML. Alpha-1-Acid Glycoprotein Quantification via Spatial Proximity Analyte Reagent Capture Luminescence Assay: Application as Diagnostic and Prognostic Marker in Serum and Effusions of Cats with Feline Infectious Peritonitis Undergoing GS-441524 Therapy. Viruses. 2024 May 16;16(5):791. doi: 10.3390/v16050791. Tršar L, Štrljič M, Svete AN, Koprivec S, Tozon N, Žel MK, Pavlin D. Evaluation of selected inflammatory markers in cats with feline infectious peritonitis before and after therapy. BMC Vet Res. 2025 May 9;21(1):330. doi: 10.1186/s12917-025-04731-x. Ceron JJ, Pardo-Marin L, Caldin M, Furlanello T, Solano-Gallego L, Tecles F, Bernal L, Baneth G, Martinez-Subiela S. Use of acute phase proteins for the clinical assessment and management of canine leishmaniosis: general recommendations. BMC Vet Res. 2018 Jun 20;14(1):196. doi: 10.1186/s12917-018-1524-y. Rossi G. Acute phase proteins in cats: Diagnostic and prognostic role, future directions, and analytical challenges. Vet Clin Pathol. 2023 Feb;52 Suppl 1:37-49. doi: 10.1111/vcp.13238. Martínez-Subiela S, Ceron JJ. Evaluation of acute phase protein indexes in dogs with leishmaniasis at diagnosis, during and after short-term treatment. Vet Med - Czech, 2005, 50(1):39-46. doi: 10.17221/5595-VETMED. Paltrinieri S. The feline acute phase reaction. Vet J. 2008 Jul;177(1):26-35. doi: 10.1016/j.tvjl.2007.06.005. Eckersall PD, Bell R. Acute phase proteins: Biomarkers of infection and inflammation in veterinary medicine. Vet J. 2010 Jul;185(1):23-7. doi: 10.1016/j.tvjl.2010.04.009. Ceron JJ, Eckersall PD, Martýnez-Subiela S. Acute phase proteins in dogs and cats: current knowledge and future perspectives. Vet Clin Pathol. 2005 Jun;34(2):85-99. doi: 10.1111/j.1939-165x.2005.tb00019.x. Shih AW, McFarlane A, Verhovsek M. Haptoglobin testing in hemolysis: measurement and interpretation. Am J Hematol. 2014 Apr;89(4):443-7. doi: 10.1002/ajh.23623. Hazuchova K, Held S, Neiger R. Usefulness of acute phase proteins in differentiating between feline infectious peritonitis and other diseases in cats with body cavity effusions. J Feline Med Surg. 2017 Aug;19(8):809- 816. doi: 10.1177/1098612X16658925. Tasker S, Addie DD, Egberink H, Hofmann-Lehmann R, Hosie MJ, Truyen U, Belák S, Boucraut-Baralon C, Frymus T, Lloret A, Marsilio F, Pennisi MG, Thiry E, Möstl K, Hartmann K. Feline Infectious Peritonitis: European Advisory Board on Cat Diseases Guidelines. Viruses. 2023 Aug 31;15(9):1847. doi: 10.3390/v15091847. Zuzzi-Krebitz AM, Buchta K, Bergmann M, Krentz D, Zwicklbauer K, Dorsch R, Wess G, Fischer A, Matiasek K, Hönl A, Fiedler S, Kolberg L, Hofmann-Lehmann R, Meli ML, Spiri AM, Helfer-Hungerbuehler AK, Felten S, Zablotski Y, Alberer M, Both UV, Hartmann K. Short Treatment of 42 Days with Oral GS-441524 Results in Equal Efficacy as the Recommended 84-Day Treatment in Cats Suffering from Feline Infectious Peritonitis with Effusion-A Prospective Randomized Controlled Study. Viruses. 2024 Jul 16;16(7):1144. doi: 10.3390/v16071144. Addie DD, Silveira C, Aston C, Brauckmann P, Covell-Ritchie J, Felstead C, Fosbery M, Gibbins C, Macaulay K, McMurrough J, Pattison E, Robertson E. Alpha-1 Acid Glycoprotein Reduction Differentiated Recovery from Remission in a Small Cohort of Cats Treated for Feline Infectious Peritonitis. Viruses. 2022 Apr 1;14(4):744. doi: 10.3390/v14040744. Romanelli P, Bertazzolo W, Prisciandaro A, Leone A, Bonfanti U, Paltrinieri S. Measurement of Feline Alpha-1 Acid Glycoprotein in Serum and Effusion Using an ELISA Method: Analytical Validation and Diagnostic Role for Feline Infectious Peritonitis. Pathogens. 2024 Mar 29;13(4):289. doi: 10.3390/pathogens13040289.

